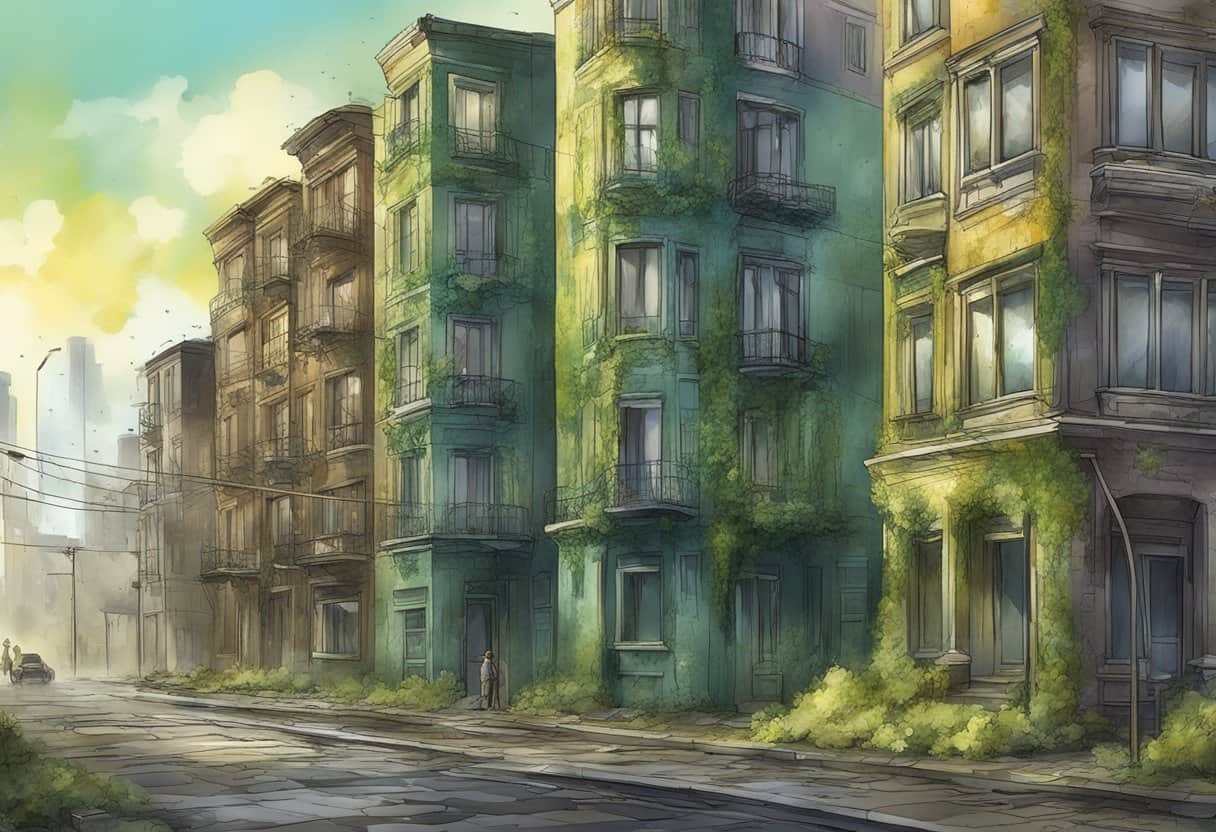Acid rain poses a significant threat to buildings, both modern and historical, around the globe. When you consider the chemical composition of acid rain, primarily a mix of sulfuric and nitric acids formed from the reaction of water vapor with sulfur dioxide (SO2) and nitrogen oxides (NOx) in the atmosphere, it’s clear why it can be so damaging. These acids fall to the earth’s surface and can corrode building materials over time. Particularly vulnerable are materials like limestone, marble, and sandstone, which often make up the splendid facades of our architectural heritage.
Understanding how acid rain affects structures requires a look into the mechanisms of material degradation. For instance, when acid rain comes into contact with calcareous stone materials, a chemical reaction occurs which can result in the loss of structural integrity and aesthetic detail, such as the erosion of intricate carvings. Beyond the visible damage, the broader ecological effects of acid rain need consideration too, as they can contribute to the long-term deterioration of buildings. With awareness and appropriate mitigation strategies, there is potential to minimize the impact of acid rain and preserve structures for future generations.
Acid rain damages buildings by corroding metals and dissolving stone, especially limestone and marble. This can lead to structural damage and loss of aesthetic detail. Protective coatings, pollution control, and using more resistant materials can help mitigate the effects.
Chemical Composition of Acid Rain

Acid rain is formed when sulfur dioxide (SO2) and nitrogen oxides (NOx) are released into the atmosphere, where they react with water, oxygen, and other chemicals to form acidic pollutants. These pollutants, primarily sulfuric acid and nitric acids, mix with clouds and precipitation to create acid rain.
Precipitation normally has a neutral pH of 7, as is seen with pure water. However, when acid rain occurs, the pH level of the precipitation drops. The pH scale, which ranges from 0 to 14, measures the acidity or alkalinity of a substance. Acid rain typically has a pH between 4.2 and 4.4, which makes it quite acidic.
- Sulfur dioxide & nitrogen oxides: Act as the primary precursors for acid rain.
- Acid precipitation: The term for rain with a pH lower than neutral.
- Formation of acids:
- SO2 combines with water to form sulfuric acid.
- NOx reacts to form nitric acid.
Even traces of carbon dioxide (CO2) in the air can contribute to the acidity of rain, albeit to a lesser extent than SO2 and NOx. Your understanding of acid rain’s impact on buildings hinges on recognizing how these chemicals transform from gaseous emissions to acid precipitation, a more tangible threat to the structural integrity of cultural and historical edifices.
Effects on Building Materials
Acid rain can have a significant impact on various building materials, leading to structural damage and aesthetic degradation over time. Your understanding of the specific reactions and effects on common materials like limestone, marble, and metals can help in better managing and preserving buildings.
Limestone and Marble Deterioration
Limestone and marble are particularly vulnerable to acid rain because they contain high levels of calcium carbonate, a substance that reacts readily with the acids present in the rainwater. This reaction often results in a loss of material, surface roughening, and increased porosity—effectively weakening the stone and leaving it more susceptible to further damage.
- Chemical equation: CaCO₃ + H₂SO₄ → CaSO₄ + CO₂ + H₂O
Sandstone and Granite Reaction
Sandstone varies in its susceptibility to acid rain, with some forms being more reactive due to their calcite content. Granite, generally resistant due to its silicate minerals, can still show signs of weathering, particularly in the form of gypsum crusts forming on surfaces, which might peel away, taking granite particles with them.
- Signs of reaction:
- Sandstone: Erosion of cementing materials, loss of carved details.
- Granite: Black crust formation, surface flaking.
Metal Corrosion
Metal components of buildings, including steel, can corrode at an accelerated rate when exposed to acid rain. This reaction not only weakens the structural integrity of the metal but also can cause staining on the building surfaces from the rust and other byproducts of the corrosion process.
- Affected metals: Primarily iron, steel, and copper.
- Result: Increased maintenance costs, potential for structural failure.
Paint Degradation
The protective paint layers on buildings are not exempt from acid rain damage. Acidic conditions can break down the chemical composition of the paint, leading to fading, cracking, and eventual peeling. This exposes underlying materials to the elements and can necessitate frequent repaintings.
- Observable effects: Color fading, chalking, and blistering.
- Long-term effect: Shortened lifespan of paintwork, increased vulnerability of the substrate.
Visible Damage to Architecture
When your community’s revered monuments and statues begin to show signs of wear, it’s often due to the silent but steady impact of acid rain. The damage can range from subtle changes to severe deterioration, all of which not only diminishes the aesthetic appeal but also the cultural significance and structural integrity of these edifices.
Surface Erosion
Acid rain actively contributes to the deterioration of exposed surfaces, especially on monuments and tombstones made of susceptible materials like limestone and marble. The removal of material over time results in the loss of carved details, directly affecting the intricate artistry that gives these structures their unique character. You might notice that once sharp inscriptions become difficult to read, or elaborate designs appear smoothed and weathered, this reveals crumbling stone where the integrity of stonework is compromised.
Black Crust Formation
Blackened crusts form on your buildings as a result of acid rain reacting with the stone in a process called sulfation. These crusts are especially prominent on protected surfaces that remain relatively dry, allowing pollutants to accumulate and react with the stone. This contrasts with exposed areas that are frequently washed away by rain, removing the crust along with minute particles of the stone itself. Over time, this cyclic process accentuates the visual contrast between washed and protected surfaces, making the impact of acid rain damage remarkably pronounced and concerning.
Impact on Cultural Heritage
Acid rain poses a significant threat to cultural heritage, including historic limestone buildings, monuments, and tombstones. You might find that these structures, made from calcareous materials, are particularly vulnerable to deterioration.
Limestone Buildings: Acid rain can accelerate the decay of building materials. Your cultural landmarks made of limestone may suffer from chemical reactions that cause the stone to weaken and crumble over time. This effect on calcareous stones is a matter of concern for the preservation of such historically valuable structures.
Monuments and Tombstones: These structures often bear the brunt of acid rain. The details and inscriptions on them can become illegible, and the physical integrity of these objects can be compromised. Notable examples include the Taj Mahal and other monuments that have experienced notable damage due to acid deposition.
National Atmospheric Deposition Program (NADP): This program tracks acid rain and its effects, including on cultural heritage. The data collected help in understanding the scope of the impact and in developing strategies to protect vulnerable structures.
When discussing the effects on lakes and the broader environment, it’s crucial to remember that the damage to inanimate objects like buildings often reflects a larger issue affecting ecosystems, which is another area monitored by programs like the NADP.
It’s important to consider measures to mitigate the effects of acid rain on cultural heritage. Protective coatings and the implementation of pollution controls are potential methods to preserve the integrity and beauty of affected structures, ensuring they remain a part of your history for generations to come.
Mechanisms of Material Degradation
When you consider the durability of buildings, it’s essential to understand that materials used in construction can degrade over time through various mechanisms caused by pollution and acid rain. These processes generally fall into two categories: chemical and physical weathering.
Chemical Weathering Processes
Chemical weathering involves the chemical reactions between the materials and the acidic compounds found in rainwater. When acids from acid deposition come into contact with the minerals in building materials, they can undergo a reaction. For example, calcium carbonate, a common mineral in limestone and marble, can react with sulphuric or nitric acids from rain to form calcium sulphate or calcium nitrate, which are more soluble in water. This increased solubility can lead to a loss of material and surface detail over time.
- Acidic Pollutants:
- Sulphur dioxide (SO2)
- Nitrogen oxides (NOx)
- Reactions:
- Calcium carbonate (CaCO3) + sulfuric acid (H2SO4) → calcium sulfate (CaSO4) + carbon dioxide (CO2) + water (H2O)
- Calcium carbonate (CaCO3) + nitric acid (HNO3) → calcium nitrate (Ca(NO3)2) + carbon dioxide (CO2) + water (H2O)
These reactions indicate how sulfuric and nitric acids, which form from atmospheric pollution, actively contribute to the deterioration of construction materials.
Physical Weathering Through Acid Deposition
Physical weathering by acid deposition, although less direct than chemical alterations, can still have significant impact. Acids and pollutants in rainwater can infiltrate porous materials, and as the water evaporates, it leaves behind salts and acidic compounds that can gradually expand and cause the material to fracture and crumble from within.
A clear example of this effect is when sulfate salts form from the aforementioned chemical reactions. These salts can crystallize within the pores of stonework leading to a form of physical weathering known as salt weathering. This process is especially destructive during cycles of wetting and drying, which can pressurize the internal structure of materials and can cause pieces of the surface to break off, a phenomenon known as scaling.
In both types of weathering, the consequences of pollution and acid rain are not just a theoretical concern but a real threat to the longevity and aesthetic of buildings and structures.
Environmental Factors Influencing Damage
Understanding how acid rain can affect buildings is crucial because environmental factors that vary from location to location determine the severity of the impact. Certain climatic conditions and the level of air pollution, especially in urban settings, are significant contributors to the damage.
Climatic Conditions
Acid rain is the result of precipitation mingling with atmospheric pollutants. The extent of damage to buildings is highly dependent on your area’s specific climate. Areas with high humidity and frequent rain events tend to experience more rapid deterioration. Seasonal variations also influence the rate at which acid rain damages structures; for example, freeze-thaw cycles can exacerbate the effects of acidic conditions on building materials.
Air Pollution and Urban Settings
In urban environments, the concentration of air pollution often amplifies the effects of acid rain. Buildings in these areas are continuously exposed to a combination of pollutants and acidic rain, leading to a greater degree of wear and corrosive damage. The presence of sulfur dioxide and nitrogen oxides—byproducts of industrial activities and vehicles—increases the acidity of rainwater. Consequently, acid rain in cities with high pollution levels can have a more severe impact on limestone and marble structures, which are particularly vulnerable to acidic conditions.
Prevention and Mitigation Strategies
The deterioration of buildings due to acid rain can be addressed through informed choices in construction materials and practices, along with broader pollution control measures. By understanding and implementing effective strategies, you can significantly reduce the impact of acid rain on building structures.
Building Material Choices
When selecting materials for your buildings, prioritize those with a higher buffering capacity to neutralize acid deposition. Use stones and metals that are less reactive to sulfuric and nitric acids. For example, granite has a greater resistance to corrosion compared to limestone or marble. Additionally, consider using aluminum for architectural details, as it forms a protective oxide layer that can resist acid damage.
Protective Coatings and Treatments
To further protect building surfaces, apply water-repellent coatings that prevent acid rain from penetrating materials. Regular treatments with sealants can also safeguard historic artifacts and sandstone structures. If you’re in an area that’s prone to heavy industrial pollution, these coatings are particularly valuable for preserving the integrity of exposed surfaces in sheltered areas.
Regulations and Emissions Reduction
Supporting legislation that leads to the reduction of fossil fuels combustion is a key step. Encourage industries to adopt cleaner technologies that reduce the emission of sulfur dioxide (SO2) and nitrogen oxides (NOx), the precursors to acid rain. You can also advocate for alternative energy solutions to decrease reliance on electricity generated from coal and other high-sulfur content fuels. Participate in community initiatives that aim to monitor and regulate human activities contributing to acid deposition, thus playing a role in protecting your local buildings and infrastructure.
Acid Rain’s Broader Ecological Effects
Acid rain is not limited to buildings; it has far-reaching impacts on the environment that you may observe in various ecosystems.
Soil: Acid rain dramatically alters the chemical balance of soil, stripping away vital nutrients and releasing aluminum, which makes it hard for plants to take up water.
Trees and Forests: The health of forests is compromised by acid rain. As acidic precipitation seeps into the soil, tree roots become damaged, and important nutrients are leached away, leaving trees weakened and susceptible to disease.
Animals: If the pH of soil and water drops, it can become toxic to animals. Birds and mammals that eat insects or plants exposed to acid rain can suffer as their food sources are depleted or poisoned.
Aquatic Environments: When acid rain flows into streams, rivers, and lakes, the consequences can be devastating:
- pH Balance: Acidic waters disrupt the pH balance, making the environment hostile for aquatic life.
- Fish Populations: Fish, like trout and salmon, are particularly sensitive to acidic conditions. Breeding and the survival of juvenile fish are hindered, leading to decreased populations.
Streams: Not only does acid rain directly impact stream water quality, but the phenomenon known as “dry deposition” can result from coal combustion byproducts settling onto land and waters surfaces, further contributing to the acidification of streams.
Remember, what you witness in local streams and forests is part of a larger pattern. The effects ripple through the food chain, leading to a domino effect that can radically alter entire biological communities. Your awareness and understanding of these broader ecological effects are crucial for addressing the challenges posed by acid rain.
Public Health and Safety Concerns
When you consider the impact of acid rain on buildings, it’s important to acknowledge the broader implications for public health and safety. Buildings degraded by acid rain can become structurally unsound, presenting direct physical hazards to inhabitants and the public. However, beyond the immediate risks of crumbling facades or weakened structures, there’s a more insidious threat to your health emanating from the same pollutants that cause acid rain.
- Sulfur dioxide (SO₂) and nitrogen oxides (NOₓ), the primary precursors to acid rain, contribute to the formation of particulate matter and ground-level ozone, contaminating the air you breathe.
- Prolonged exposure to these polluted air particles can exacerbate respiratory conditions, such as asthma, and lead to chronic health problems.
- Your respiratory system can become irritated, decreasing lung function and making breathing more difficult.
Furthermore, buildings tainted with acidic substances can release particles into the air during degradation. If these fine particles are inhaled, they may pose additional health risks. Here’s a quick rundown of the components affecting the air:
| Pollutant | Health Impact |
|---|---|
| Sulfur dioxide (SO₂) | Respiratory irritation, asthma aggravation |
| Nitrogen oxides (NOₓ) | Lung problems, lower immunity to infections |
Being aware of these health concerns, you should support regular monitoring of air quality, especially if you live in areas prone to the effects of acid rain. Adequate building maintenance and restoration work help mitigate the falling debris risks, while air quality improvements can reduce the secondary health effects linked to the substances that cause acid rain.
Historical and Contemporaneous Case Studies
When you explore the impact of acid rain on buildings, you might consider examples from Europe, particularly in England, where the effects on stone surface materials are quite evident. A notable instance is the mutable stone of St Ann’s Church, Manchester, where the intricate interplay of the stone’s response to atmospheric conditions has been documented. The sulphuric acid and nitrates present in the atmosphere contribute significantly to this type of weathering.
- Roughened Surfaces: Acid rain primarily affects building materials that are composed of calcium carbonate, such as limestone and marble, leading to a roughening of surfaces.
- Chemical Reactions: This weathering is a result of a chemical reaction where acidic pollutants in the air convert to acids when mixed with water, which then dissolve the stone material.
In your review of historical studies, you would find indications of how the lack of understanding of these processes in the past contributed to the decline in structural integrity of the buildings affected. Contemporary analyses can provide insight into how these materials might continue to deteriorate, leading to efforts for better preservation.
The National Atmospheric Deposition Program in the United States is one such initiative that tracks the deposition of pollutants that contribute to acid rain. It offers a broad perspective on the relationships between emissions, deposition, and effects on materials, including stone surfaces on historical buildings. Understanding these relationships is key to formulating strategies to protect prized architectural heritage.
Monitoring and Research
To ensure that you can fully grasp the impact of acid rain on buildings, it’s crucial to understand the role of monitoring and research activities. Agencies like the National Atmospheric Deposition Program (NADP) work tirelessly to collect and analyze rainfall data which provides scientists with critical insights into the composition of precipitation and its corrosive effects
Elements such as sulfur dioxide and nitrogen oxides from both natural sources and human activities contribute to the formation of acid rain. Researchers analyze these components to predict and mitigate potential damage.
In the realm of climate change, it’s understood that weather patterns might shift, potentially altering the frequency and intensity of acid rain events. Your comprehension of this relationship is vital, as it helps scientists to model and anticipate changes in acid rain’s effects on buildings.
Here is how you can stay informed on the research:
- Review Reports: Regular publications from research entities give updates on the latest findings.
- Participation in Surveys: Contributing to surveys helps collect data on a broad scale.
- Understanding the Data:
- pH levels: Determining the acidity of rainwater.
- Concentration of acidic elements: Checking for sulfuric and nitric acids.
Monitoring involves using specialized equipment to measure the impact over time, which may include observing changes in:
- Building Materials: Identifying which types are most vulnerable.
- Heritage Structures: Assessing the risk factors for historical buildings.
Frequently Asked Questions
Acid rain has a significant impact on buildings, leading to structural damage and deterioration over time. Uncover the specific ways acid rain interacts with different building materials and learn about the most effective measures to mitigate its effects.
What types of materials used in building construction are most vulnerable to acid rain damage?
Materials such as limestone, marble, and certain sands containing carbonate compounds are highly susceptible to acid rain damage. These materials can undergo a chemical reaction leading to their dissolution when exposed to acidic conditions.
What are the visible signs of acid rain deterioration on buildings?
Visible signs include the loss of surface material, an increase in surface roughness, and changes in color or appearance of the building facade. Acid rain may cause certain materials to weaken, leading to pitting, flaking, and scaling on the building stones.
How does acid rain contribute to the weathering and erosion of building structures?
Acid rain significantly accelerates the natural processes of weathering and erosion by chemically reacting with the minerals in building materials. Over time, this reaction breaks down the structural integrity, causing pieces to fall away and surfaces to dissolve.
In what ways can acid rain impact historical monuments and landmarks?
Historical monuments and landmarks, often constructed with vulnerable materials like marble and limestone, face degradation from acid rain. This can lead to a loss of intricate details, inscriptions, and even structural failure of culturally significant sites.
Which preventive measures can protect buildings from the effects of acid rain?
Preventive measures include applying water-repellent coatings to vulnerable surfaces, using acid-resistant materials during construction, and implementing pollution reduction techniques to lower the overall acidity of rainwater.
How does the chemical reaction between acid rain and building materials occur?
The chemical reaction typically involves the dissolution of carbonate compounds found in many building materials when they react with the sulfuric and nitric acids present in acid rain. This chemical reaction can result in the release of carbon dioxide, water, and dissolved mineral residues from the building’s materials.